Mastering Calcium Hardness in Pool Water
Understanding the Importance and Management of Calcium Hardness for a Healthy Swimming Environment
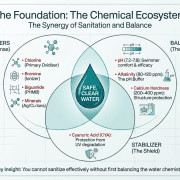
Calcium hardness is a crucial aspect of pool water chemistry that is often overlooked. It refers to the amount of calcium dissolved in the pool water and plays a significant role in maintaining the overall water balance. In this article, we will discuss what calcium hardness is, why it’s important, and how to manage it effectively to ensure a safe and enjoyable swimming experience.
What is Calcium Hardness?
Calcium hardness is a measure of the concentration of calcium ions (Ca2+) in the pool water. It is typically expressed in parts per million (ppm) and is an essential component of the water’s overall mineral content. Calcium is a naturally occurring element that can be found in most water sources, including tap water and groundwater. In swimming pools, calcium hardness can be affected by various factors, such as the water source, the pool’s construction materials, and the chemicals used for maintenance.
Why is Calcium Hardness Important?
- Protecting Pool Surfaces and Equipment: Maintaining proper calcium hardness levels helps protect the pool’s surfaces, equipment, and plumbing system from damage. Low calcium hardness can cause the water to become corrosive, potentially leading to etching and staining of pool surfaces, as well as damage to metal fixtures and equipment. Conversely, high calcium hardness can cause scaling, which can clog filters, reduce circulation, and lead to unsightly deposits on pool surfaces.
- Water Balance: Calcium hardness plays a crucial role in the overall water balance of the pool. It is directly related to the pool’s pH and alkalinity levels, and maintaining appropriate calcium hardness levels is necessary to prevent fluctuations in these parameters.
- Swimmer Comfort: Proper calcium hardness levels contribute to a more comfortable swimming environment by helping to prevent eye and skin irritation.
Managing Calcium Hardness
- Test the Water: Regularly test your pool water for calcium hardness using a reliable test kit. The ideal calcium hardness level for a swimming pool is between 200 and 400 ppm. However, some pool finishes may require slightly different levels, so consult your pool manufacturer’s guidelines for specific recommendations.
- Adjust Calcium Hardness Levels: If your pool’s calcium hardness is too low, you can raise it by adding calcium chloride, following the manufacturer’s instructions. If the calcium hardness is too high, you can lower it by diluting the pool water with fresh water, using a flocculant to help remove excess calcium, or using a specialized calcium-reducing product.
- Maintain Water Balance: To prevent calcium hardness fluctuations, it is essential to maintain proper water balance. Test and adjust the pool’s pH, alkalinity, and sanitizer levels regularly.
- Prevent Scaling and Staining: To minimize the risk of scaling and staining, avoid using calcium-based pool chemicals, such as calcium hypochlorite, if your pool’s calcium hardness is already high. Use a sequestering agent or stain inhibitor to help prevent calcium deposits and protect pool surfaces.
- Regular Maintenance: Regularly brush and vacuum your pool to remove any buildup of calcium deposits, and maintain your pool’s filtration system to ensure efficient circulation and water balance.
Calcium hardness is a vital aspect of pool water chemistry that is crucial for protecting pool surfaces, equipment, and ensuring a comfortable swimming environment. By understanding what calcium hardness is, its importance, and how to manage it effectively, pool owners can maintain a well-balanced and healthy swimming pool. Regular testing and adjustments, as well as preventative measures, are the keys to success in mastering calcium hardness in your pool.
Recommended Products
- Taylor K-2006 Complete Test Kit
- AquaChek 7-Way Test Strips
- In The Swim 3-Inch Chlorine Tablets
- In The Swim Chlorine Pool Shock
- Liquid Chlorine
- Clorox Pool&Spa pH Down
As an Amazon Associate, we earn from qualifying purchases.